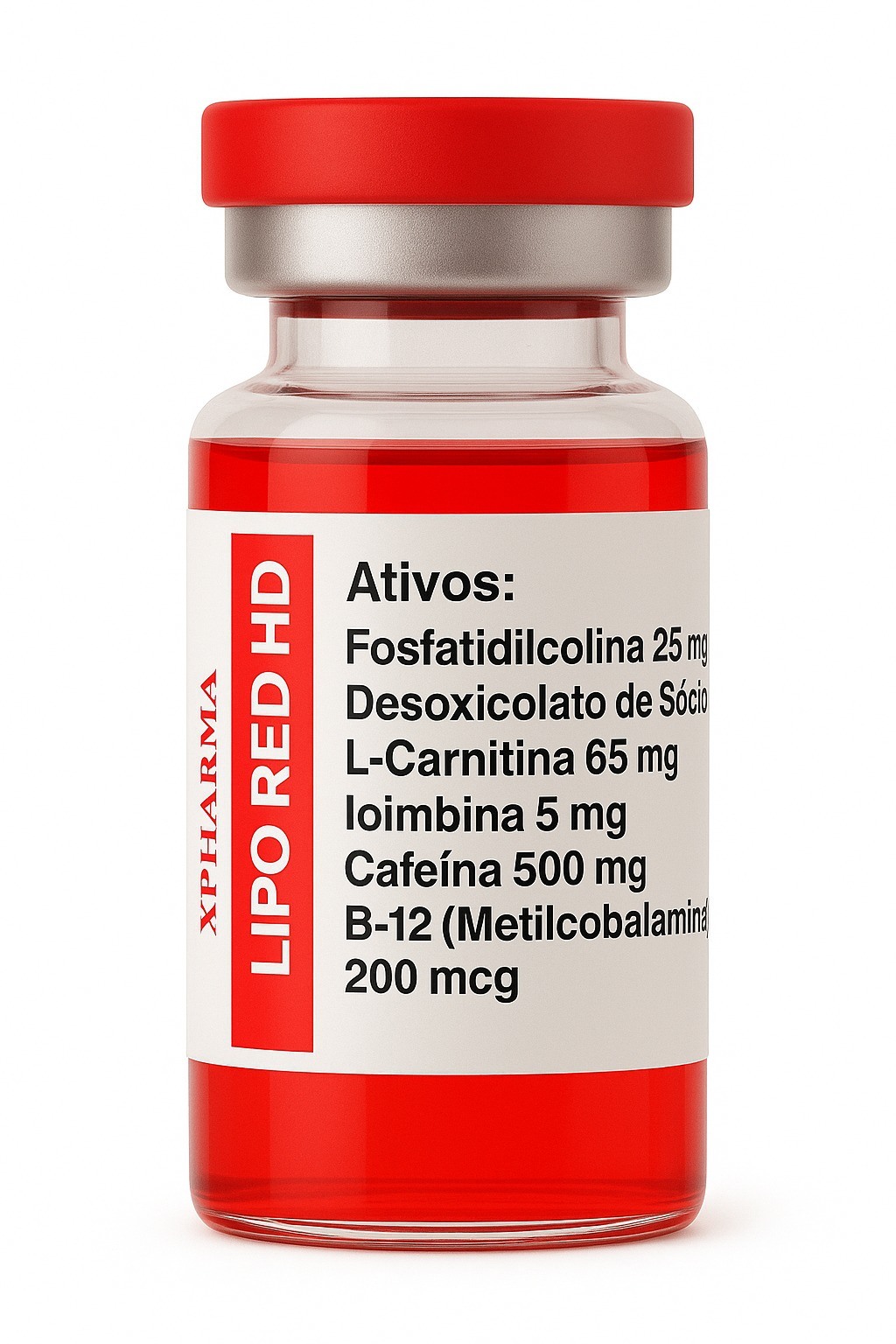
LipoRED HD
Product presentation from the XPharma Premium line. Commercial information available upon request.
XPharma presents premium product lines built for institutional review, commercial documentation, and official product information channels.

XPharma was built to support premium product presentation, disciplined quality control, and institutional communication aligned with recognized manufacturing standards.
Premium product presentations designed for institutional review, commercial inquiries, and official product information channels.
From product presentation to commercial documentation, every detail is built around clarity, consistency, and quality control.
Process discipline, documentation review, and product-specific quality information through official XPharma channels.
Institutional materials are organized for product evaluation, commercial due diligence, and market-specific documentation.
Clear labeling, organized product information, and official communication standards support responsible commercial review.
Our team provides commercial and product information for professional, distributor, and multi-market institutional inquiries.
Product documentation, quality information, and market-specific requirements may vary by product and commercial context. Official product information is available upon request.
Product documentation may be provided through official XPharma channels, when applicable.
Quality process information may be reviewed by product, market, and commercial context.
Market-specific documentation should be reviewed before commercial distribution.
Commercial information and product details are available through official channels.
Operational information is organized for responsible documentation review and official commercial follow-up.
Information about XPharma product lines, presentations, and commercial channels is centralized on the institutional pages of this site. For specific questions, contact our team through official channels.
Every catalog reference is organized to support product identification, documentation review, label consistency, and official commercial follow-up. Details may vary by product line and market context, and should be confirmed through XPharma official channels.
Clarity, credibility, and responsible documentation are part of the XPharma standard.
Distributors, clinics, and professional buyers can request product information, commercial details, and market-specific documentation from the XPharma team.